Cultivated meat is here, but policies decide how fast it reaches you. Singapore and the US lead the way, already approving several products, while regions like the EU and UK are still navigating slower processes. Key factors influencing availability include:
- Regulations: Countries like Singapore have streamlined approval systems, while the EU faces delays with its two-step process.
- Labelling: Terms like "cell-cultivated" are becoming standard in the US, but rules vary globally.
- Funding: Governments have invested £2 billion globally in alternative proteins, but it's far from the £8 billion needed annually.
- Trade Rules: Restrictions and bans in some areas, like Italy, complicate market access.
Early approval markets like Singapore and the US are gathering data and refining processes, but for regions like the UK, cultivated meat for human consumption is still years away. Policies around funding, labelling, and trade will ultimately dictate how soon these products become a regular choice for consumers.
Webinar_Navigating the regulatory landscape of cultivated meat across regions (17 April 2026)
sbb-itb-c323ed3
Global Regulatory Pathways: How Different Regions Approve Cultivated Meat
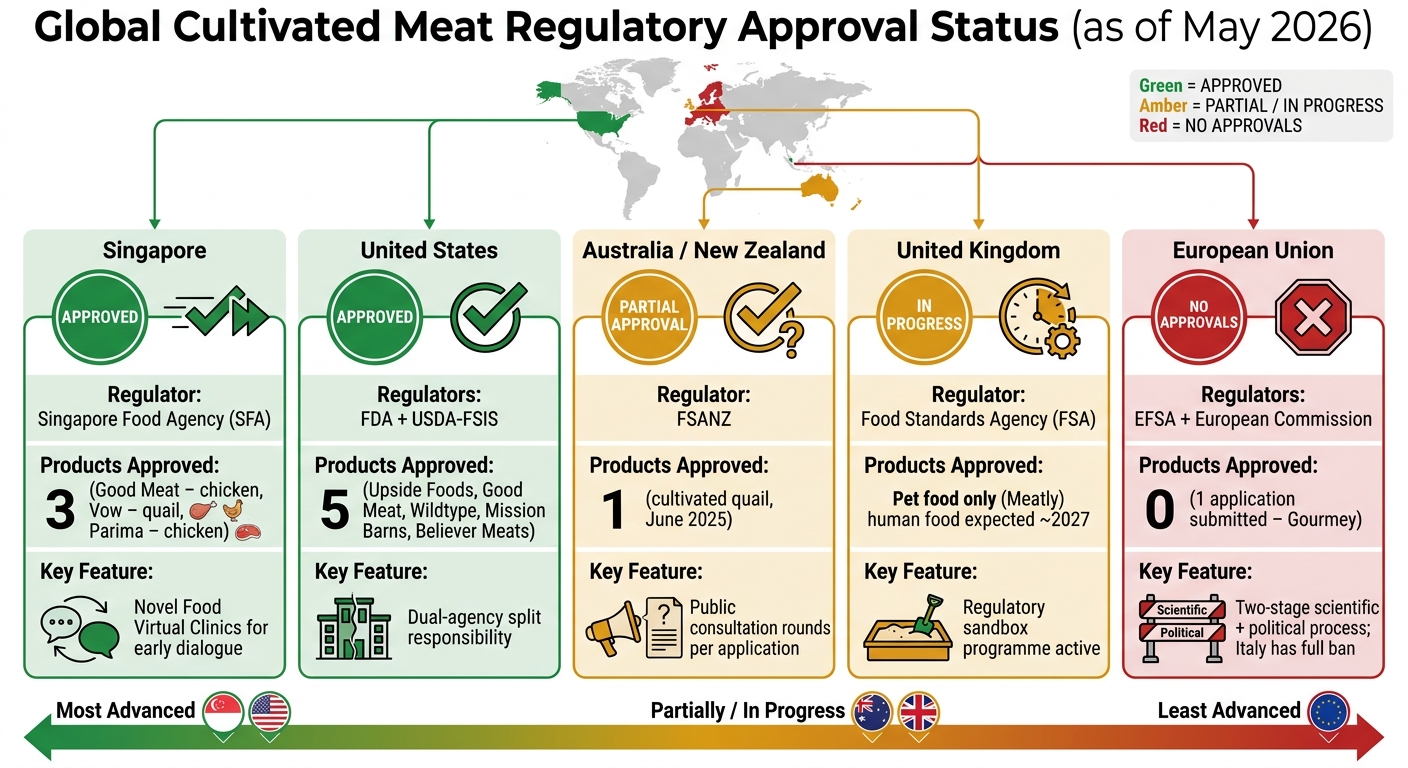
Global Cultivated Meat Regulatory Approval Status 2026
Getting Cultivated Meat to market requires navigating a maze of regulations that differ from country to country. Each region has its own food safety standards, creating a patchwork of approval processes and timelines.
Early Movers: Singapore, the United States, and Australia/New Zealand
Singapore was the first country to greenlight Cultivated Meat for human consumption, cementing its reputation as a forward-thinking hub. The Singapore Food Agency (SFA) encourages early conversations between regulators and producers through "Novel Food Virtual Clinics." These sessions allow companies to discuss their products before submitting formal applications. By early 2026, three products had been approved: Good Meat (chicken), Vow (quail), and Parima (chicken) [2]. In January 2025, Singapore introduced the Food Safety and Security Act (FSSA), which established a clear legal framework for foods produced using animal cell culture [3]. This proactive approach has sped up the timeline for getting products to consumers.
In the United States, the approval process is split between two agencies: the FDA, which handles pre-market consultations and cell bank safety, and the USDA-FSIS, responsible for processing and labelling. This division of duties has enabled five products to secure approval: Upside Foods, Good Meat, Wildtype, Mission Barns, and Believer Meats [2]. In June 2023, after completing FDA reviews and USDA inspections, Upside Foods and Good Meat began selling Cultivated chicken at restaurants in San Francisco and Washington D.C. [3].
Australia and New Zealand, overseen by FSANZ (Food Standards Australia New Zealand), add a unique layer to their process: public consultation. This allows citizens to provide input on specific product applications. For instance, in June 2025, the Australia New Zealand Food Standards Code was updated to approve cell-cultured quail as a novel food ingredient. This followed a 2023 submission by Vow Group Pty Ltd, which underwent two rounds of public feedback before receiving the green light [3].
"The US and Singapore, no surprise there, usually lead the way... Australia and New Zealand have completed safety reviews comparable in rigour to the US." - Suzi Gerber, Executive Director, Association for Meat, Poultry and Seafood Innovation (AMPS) [2]
The EU and UK: Slower but Structured Approval Processes
The European Union bases its approval process on the Novel Food Regulation (EU) 2015/2283, which involves two main steps: a scientific risk assessment by the European Food Safety Authority (EFSA) and a political vote by member states in the Standing Committee on Plants, Animals, Food and Feed (PAFF). While the official timeline is 18 months, delays can extend this to three years due to "stop-the-clock" requests [5]. As of early 2026, only one application - from French company Gourmey - had been submitted, with no products yet approved for human consumption [5]. Political hurdles add further complications. For example, Italy passed a law in December 2023 banning the production and sale of Cultivated Meat altogether [5].
Post-Brexit, the UK has taken a different route. The Food Standards Agency (FSA) introduced a regulatory sandbox specifically for cell-cultivated products. This initiative allows for controlled testing and direct collaboration with the industry, an approach the EU has rejected under its proposed Biotech Act [4]. While the UK has approved Cultivated Meat for pet food (under the brand Meatly), human food approval is anticipated by 2027 [2][4].
"Where approval pathways are clearly defined, capital can price risk more efficiently. Where they are ambiguous or protracted, the investor base narrows to higher-risk equity." - Vicky Grinnell, Food Tech and Futures Lead, Lloyds Corporate and International [4]
Comparing Regulatory Approaches Across Regions
The regulatory frameworks in different regions significantly impact when consumers can expect to see Cultivated Meat on their shelves.
| Region | Primary Regulator(s) | Approval Status (as of May 2026) | Key Feature |
|---|---|---|---|
| Singapore | Singapore Food Agency (SFA) | 3 products approved (chicken, quail) | Early dialogue & "Virtual Clinics" |
| United States | FDA & USDA-FSIS | 5 products cleared (chicken, salmon, pork fat) | Clear division of agency responsibilities |
| Australia / NZ | FSANZ | Quail approved (June 2025) | Public consultation on individual applications |
| United Kingdom | Food Standards Agency (FSA) | Pet food approved; human food expected ~2027 | Active regulatory sandbox for cell-cultivated products |
| European Union | EFSA / European Commission | No approvals yet | Two-stage scientific and political process |
Even in regions where products have been approved, most companies are starting with small-scale restaurant launches to gauge consumer interest and refine production. Regulatory approval is just one piece of the puzzle; factors like government support, trade policies, and labelling standards also play a crucial role in determining how quickly Cultivated Meat becomes widely available [2]. This patchwork of approaches continues to shape the global conversation around market readiness and consumer access.
Beyond Regulation: Funding, Labelling, and Trade Policy
Regulatory approval is just the starting line for Cultivated Meat's journey to market. Its broader success depends on three key policy tools: public funding, labelling rules, and trade access. Each plays a crucial role in shaping how quickly and effectively this industry can grow.
Government Funding and Infrastructure Support
Governments worldwide are stepping up their investment in Cultivated Meat to drive innovation and scale production. By 2025, public funding for alternative proteins, including Cultivated Meat, reached a global total of $2.5 billion [7]. The number of countries actively supporting these policies has more than doubled, growing from 16 in 2021 to 33 by 2025 [7].
A significant portion of this funding is focused on building biomanufacturing capacity - facilities designed to grow meat cells efficiently on a large scale. Countries like Australia, Canada, South Korea, and India are encouraging the development of shared biomanufacturing hubs. These hubs aim to support local startups, reduce dependency on imports, and strengthen domestic competitiveness [7]. In Europe, the EU allocated over €100 million in loans to plant-based and fermentation companies in 2025 to help scale up production [7]. On top of that, public investment is helping to lower production costs by advancing research into serum-free growth media.
However, there’s still a long way to go. Experts estimate that $10.1 billion in annual global government spending is needed to fully realise the economic and environmental potential of Cultivated Meat. Current funding levels only cover about 10% of that benchmark [7].
As funding grows, the next challenge is ensuring consumers trust these products - starting with clear and consistent labelling.
Labelling Rules and What They Mean for Consumers
Labelling regulations are a powerful tool for building consumer confidence and shaping perceptions. Even before a product is tasted, its label can influence trust and acceptance, making these rules essential for market success.
In the United States, the USDA has taken a clear stance. As of March 2025, labels must use terms like "cell-cultivated" or "cell-cultured" directly before the product name - for instance, "cell-cultivated chicken" [6]. UPSIDE Foods was the first company to navigate this process, receiving USDA label approval and a Grant of Inspection (GOI) in 2023 after a regulatory journey that started in 2019 [6]. Approved Cultivated Meat products also carry the USDA inspection seal, signalling they meet the same federal safety standards as conventional meat.
Elsewhere, approaches vary. The UK is developing its labelling standards through a regulatory sandbox programme, while the EU handles labelling on a case-by-case basis during product authorisations [2][5]. This patchwork of rules creates hurdles for producers trying to enter multiple markets and leaves consumers with inconsistent information about what they’re buying.
| Region | Labelling Terminology | Oversight Body |
|---|---|---|
| United States | "cell-cultivated" or "cell-cultured" | USDA-FSIS and FDA [6] |
| Singapore | Product-specific (e.g., "cultivated chicken") | Singapore Food Agency [2] |
| European Union | Determined per authorisation | EFSA (Novel Food Framework) [5] |
| United Kingdom | Under development (Sandbox programme) | Food Standards Agency (FSA) [2] |
While labelling builds trust, trade policies decide how far these products can travel.
Trade and Market Access Rules
Trade policies are becoming a major factor in determining where Cultivated Meat can be sold. Some regions have outright bans, complicating market integration and potentially clashing with broader trade rules, such as those governing the EU internal market [3][5]. In the United States, states like Florida, Arizona, and Texas have introduced labelling laws that align with federal guidelines, adding further layers of complexity for producers operating across state lines [6].
The global landscape in 2025 has shifted towards prioritising domestic food security. Governments are re-shoring food production to reduce reliance on imports. For example, Singapore's Food Safety and Security Act (FSSA) of 2025 explicitly governs both imported and exported novel foods, reflecting the nation’s focus on controlling its food supply [3].
Looking ahead, reciprocal safety recognition could simplify market access. This system would allow products approved in one country to gain easier entry into others. However, such agreements will likely depend on independent reviews by major regulators [2]. Until then, producers must navigate the expensive and time-consuming process of seeking approval in each market individually, slowing the global rollout of Cultivated Meat.
Monitoring and Compliance: How Safety Is Maintained
Getting approval is just the beginning; keeping cultivated meat products safe on shelves requires strict systems that continue well after they hit the market.
Post-Market Surveillance and Traceability
Once cultivated meat products are available to consumers, maintaining safety depends on a seamless collaboration between agencies. In the United States, the FDA oversees the early stages, such as cell collection and banking, while the USDA-FSIS takes over during harvesting, processing, packaging, and labelling. A good example of this process in action is Upside Foods, which received USDA-FSIS approval in 2023 [3].
Traceability is especially critical for cultivated meat. It's not just about tracking the source animal; every component, from growth media to scaffolding materials, must be monitored. This is achieved through systems like HACCP and GMP protocols [1][2]. Advanced tools, including AI-driven inspections and automated audits, now handle these intricate supply chains in real time [1]. These measures not only ensure cultivated meat safety and consumer confidence but also align domestic practices with global expectations.
"Those who invest in clean data structures early on will be ahead of the curve." - Hensgen, QS Expert [1]
Such robust domestic systems are setting the stage for greater international collaboration.
International Coordination and Standard-Setting
To achieve consistency worldwide, international cooperation is essential. The Codex Alimentarius Commission (CAC) - a joint initiative by the FAO and WHO - addressed cultivated meat safety during its 45th session in late 2022. The frameworks developed during this session are now serving as references for national regulators as they establish their own guidelines [8]. In the EU, for instance, once a cultivated meat product is added to the Union List under the Novel Foods Regulation, it becomes subject to mandatory post-market monitoring, ensuring uniform safety measures across all Member States [9].
Progress toward global harmonisation is expected to be gradual. Suzi Gerber, Executive Director of the Association for Meat, Poultry and Seafood Innovation (AMPS), expressed optimism about this process:
"Historically, we've seen reciprocity develop once countries are comfortable managing technologies domestically. I would expect that to happen here too, eventually." [2]
For now, the focus remains on fine-tuning domestic frameworks. This cautious approach helps build the trust needed for future international integration.
What Current Policies Mean for Future Consumer Access
Early Approval Markets as Test Cases
Singapore and the US have taken the lead in setting early benchmarks for cultivated meat approval. Singapore became the first country to approve cultivated meat for human consumption and reaffirmed its commitment with the introduction of the Food Safety and Security Act (FSSA) in January 2025. Meanwhile, the US adopted a dual-agency approach, with the FDA and USDA clearing five cultivated meat products by early 2026 [2][3]. These early markets are not just paving the way for regulatory frameworks; they’re also gathering crucial data on safety, consumer preferences, and pricing. This information can serve as a valuable resource for regulators in other regions, including the UK's Food Standards Agency (FSA).
Suzi Gerber, Executive Director of the Association for Meat, Poultry and Seafood Innovation (AMPS), highlighted the significance of these early movers:
"It's an interesting time as the standard countries that usually lead the way in food safety are doing their independent safety review processes. The US and Singapore, no surprise there, usually lead the way." [2]
Progress isn’t limited to these two nations. Australia and New Zealand have also moved forward, amending the Australia New Zealand Food Standards Code in June 2025 to approve cultivated quail as a novel food ingredient, following an application by Vow [3]. These approvals are creating a foundation of evidence that could simplify and speed up the review process for countries with slower timelines.
The lessons learned from these pioneering markets are proving invaluable for policymakers in regions where approval processes take longer.
How Policy Speed and Coordination Affect Market Timelines
For UK consumers, the wait for cultivated meat remains lengthy. Despite ongoing efforts, products for human consumption are still years away from hitting retail shelves. The FSA, responsible for authorising new food products, takes an average of 2.5 years to complete approvals - far exceeding its statutory target of 17 months. Resource constraints are a key factor, with at least four cultivated meat applications under review as of late 2024 [10].
In an effort to address these challenges, the UK Department for Science, Innovation and Technology (DSIT) launched a £1.6 million regulatory sandbox in late 2024. Companies like Hoxton Farms (UK), Mosa Meat (Netherlands), and BlueNaju (USA) are participating, focusing on creating guidance for hygiene and cultivated meat labels [10]. Dr Max Jamilly, CEO of Hoxton Farms, commented on the initiative:
"The FSA has made great steps with the £1.6 million in funding for the sandbox, but it will need more resources to keep up the pace. The FSA is managing a high volume of novel food applications across multiple categories, which has stretched its capacity and slowed approvals." [11]
Interestingly, the UK has made quicker progress in the pet food sector. In July 2024, Meatly successfully navigated a fast-track review process for cultivated pet food, giving the UK an early lead in this niche market. This demonstrates how alternative regulatory pathways can help accelerate product availability in specific categories [10].
Helping Consumers Prepare for What Is Coming
As regulatory frameworks continue to evolve, it’s essential to keep consumers informed and ready for what’s ahead. The speed of policy development directly impacts when UK shoppers will be able to purchase cultivated meat. Platforms like Cultivated Meat Shop aim to bridge this gap, offering UK and European consumers clear, up-to-date information about cultivated meat technology, products in development, and how regulations are shaping future availability. For those eager to stay ahead, the site even offers a waitlist to ensure they’re among the first to know when products hit the UK market.
Conclusion: Policy as the Gateway to Consumer Access
Regulatory approval is essential for consumer access, but it’s only the first step. Even after receiving clearance, products often start in select restaurant settings rather than hitting supermarket shelves. This allows companies to fine-tune pricing, assess consumer interest, and scale production. Take the case of Believer Meats: despite earning regulatory approval in the US, the company shut down in December 2025 due to the long, costly journey from approval to widespread availability [2].
While progress varies, it is undeniably gaining momentum. Early adopters like Singapore and the US demonstrate how clear policies can speed up product approvals. Meanwhile, Australia and New Zealand updated their food standards code in June 2025, offering a stronger foundation for faster and more informed regulatory decisions. These advancements not only boost investor confidence but also lay the groundwork for broader consumer access.
Policy clarity is shaping market evolution, but consumer readiness is equally critical. Labelling, trade agreements, funding, and post-market monitoring all play a role in market development. However, it is the efficiency and transparency of regulatory frameworks that will dictate how quickly cultivated meat moves from being a niche product to a common choice for UK shoppers. As Vicky Grinnell, Food Tech and Futures Lead at Lloyds Corporate and International, explains:
"Where approval pathways are clearly defined, capital can price risk more efficiently. Where they are ambiguous or protracted, the investor base narrows to higher-risk equity." [4]
The ultimate aim, as Suzi Gerber of AMPS puts it, is simple: "We want the public to feel confident that what they're eating is not just safe, but held to the highest safety standard of any food on the market." [2]
As policies continue to evolve, staying informed is vital. Cultivated Meat Shop keeps track of developments in major markets, helping UK consumers understand the current landscape and what lies ahead. They even offer waitlists for early access when products become available locally, ensuring shoppers are prepared for the changes on the horizon.
FAQs
Why is Cultivated Meat approved in some countries but not others?
Regulatory approval for cultivated meat differs widely across the globe, shaped by varying frameworks, safety assessments, and policy approaches. Take Singapore, for example - it has taken the lead with well-defined approval processes, paving the way for cultivated meat products to reach consumers. Meanwhile, regions like the EU are still fine-tuning their pathways under the Novel Food Regulation, which slows things down.
In the US, federal approval is in place, but state-level regulations can sometimes complicate matters. Similarly, in areas like the UK and the EU, progress is hindered not just by regulatory uncertainty but also by the high costs of production, which remain a significant barrier to widespread adoption.
What does the UK need to change to approve cultivated meat sooner?
To accelerate the approval process for cultivated meat in the UK, the regulatory framework needs adjustments. Key efforts include the Food Standards Agency's ongoing safety review and the £1.6 million sandbox programme designed to explore innovative approaches. Aligning UK regulations with international standards, cutting unnecessary red tape, and boosting public funding for research are also critical steps. These measures aim to shorten approval timelines and enable scalable, cost-efficient production, helping cultivated meat reach consumers faster.
How will safety be monitored after cultivated meat goes on sale?
Once cultivated meat hits the shelves, its safety will be closely monitored by government regulatory bodies. These agencies will handle licensing, oversight, and post-market checks to ensure everything stays on track. Countries such as Singapore, the United States, and the United Kingdom are already working on frameworks to guarantee safety. Key measures like continuous testing, traceability systems, and regular reporting will play a crucial role in identifying and addressing any potential risks, helping cultivated meat become a reliable alternative to traditional meat.













